タイトルまたは説明
Divalproex extended-release tablets Pronunciation
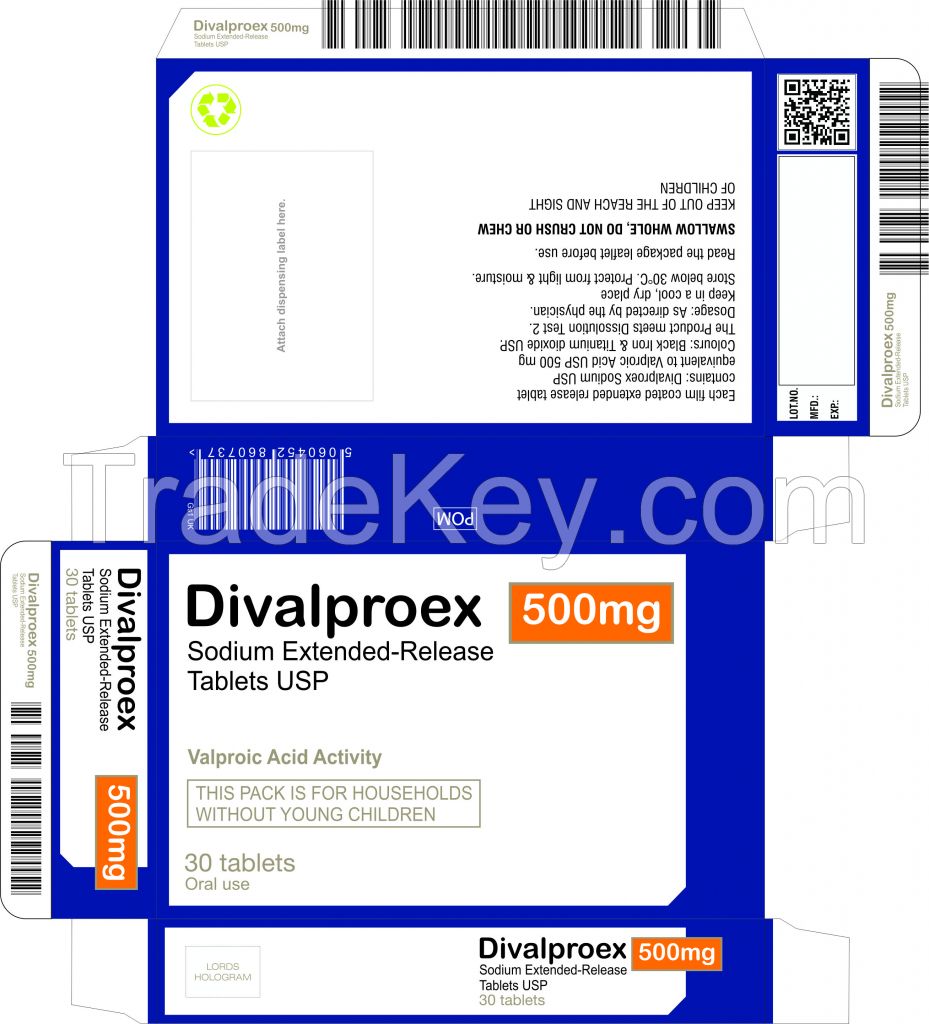
Generic Name: divalproex (dye-VAL-proe-ex)
Brand Name: Depakote ER
Liver failure and death from liver failure has occurred in patients
taking divalproex extended-release tablets. This has usually
occurred within the first 6 months of treatment. Tell your doctor
right away if you notice symptoms of liver problems (eg, a general
feeling of discomfort, sluggishness, unusual tiredness or weakness,
swelling of the face, loss of appetite, vomiting, stomach pain,
dark urine, pale stools, or yellowing of the skin or eyes). In
patients who have seizures, loss of seizure control may occur. You
should have lab tests done before and during treatment to check for
liver problems. Be sure to keep all doctor and lab
appointments.
Children younger than 2 years old have an increased risk of fatal
liver problems, especially if they take more than 1 seizure
medicine or have a metabolic disorder, a severe seizure disorder
along with mental retardation, or organic brain disease. Discuss
any questions or concerns with your doctor.
There is an increased risk of liver failure and death from liver
failure in patients who have a genetic liver problem caused by a
mitochondrial disorder (eg, Alpers-Huttenlocher syndrome). You may
need to have a special genetic test to check for this condition.
Certain patients who have known or suspected mitochondrial
disorders should not take divalproex extended-release tablets. Talk
with your doctor for more information.
Divalproex extended-release tablets can cause severe birth defects
if it is used during pregnancy. It can also cause the child to have
a lower IQ. Do not take divalproex extended-release tablets to
prevent migraine headaches if you are pregnant. If you are pregnant
and take divalproex extended-release tablets for seizures or
bipolar disorder, talk to your doctor to decide if you will
continue to take divalproex extended-release tablets.
If you are able to become pregnant, you must use an effective form
of birth control while you take divalproex extended-release
tablets. Contact your doctor right away if you become pregnant or
think you may be pregnant while taking divalproex extended-release
tablets.
Divalproex extended-release tablets comes with an extra patient
information sheet called a Medication Guide. Read it carefully.
Read it again each time you get divalproex extended-release tablets
refilled.
Severe and sometimes fatal pancreas problems (pancreatitis) have
occurred with the use of divalproex extended-release tablets. This
has been reported shortly after starting treatment as well as after
several years of use. Seek immediate medical attention if you
notice any stomach pain, nausea, vomiting, or loss of appetite.
Divalproex extended-release tablets are used for:
Controlling certain types of seizures in the treatment of epilepsy.
It is also used to treat the manic phase of bipolar disorder
(manic-depressive illness) and prevent migraine headaches. It may
also be used for other conditions as determined by your doctor.
Divalproex extended-release tablets are an anticonvulsant. It works
by reducing or preventing the number of seizures by controlling the
abnormal activity of nerve impulses in the brain and central
nervous system. Exactly how it works to treat bipolar disorder and
migraine headache is not known.
Do NOT use divalproex extended-release tablets if:
you are allergic to any ingredient in divalproex extended-release
tablets
you have liver problems or a urea cycle disorder
you have a genetic liver problem caused by a mitochondrial disorder
(eg, Alpers-Huttenlocher syndrome)
the patient is younger than 2 years old and has a mitochondrial
disorder
you are taking divalproex extended-release tablets to prevent
migraine headaches and you are pregnant
Contact your doctor or health care provider right away if any of
these apply to you.
Advertisement
SLIDESHOW
Epilepsy in Adults: A Healthcare Professional's Guide
Before using divalproex extended-release
FDA approval given for Divalproex to Indian Drug Company Taj
Pharmaceuticals 5th February ***8, Indian Pharmaceuticals Company
Taj Pharmaceuticals based in Mumbai announced FDA approval for
Divalproex Generic drug manufacturing for regulatory market and
non-regulated market. The company is said to have approvals for
different combinations of the drug, i.e. Divalproex sodium **0mg,
Divalproex sodium **0mg, Divalproex sodium **0mg ER, Divalproex
sodium **0mg ER, Divalproex sodium **5mg, Divalproex sodium **0mg,
Divalproex sodium ***0mg, Divalproex sodium 1g, Divalproex sodium
**0mg ER . The generic manufacturing strategies of New Drug
Divalproex and the product-process of Taj Pharmaceuticals Ltd. are
compared and collaborated using data from global Pharmaceuticals
manufacturing plants in the UK(Newcastle), US, Korea, Italy,
Germany and Japan. The Indian company is eminent manufacturer,
supplier and exporter of Divalproex and various pharmaceuticals
generics medicines in Tablets, capsules, Vial (injections), ampule,
Strips and schedule packs etc. The companies integrated research
team facilitates the affordable production of the drug for even
non-regulated markets of the world. i.e. Middle East, Thailand,
Bangladesh etc. The company is also involved in manufacturing of
Active Ingredient of various pharmaceuticals finished formulations
in IP /BP / USP grades for all markets. The approvals bring a
positive uplift in the share market and public perception for
Pharmaceuticals investors. Taj Pharmaceuticals Limited Working For
Healthier World INDIA FOR IMMEDIATE RELEASE Mumbai - 5th February
***8 - Drug firm Taj Pharmaceuticals today said it has received
tentative approval from Food and Drug Administration regulator to
sell a generic version of Divalproex Taj Pharma Group announces U
SF D A t ent a t i v e ap p r o v a l f o r Va r i ous g ene r i c
M ed i c ine s Note: This site contains medical information that is
intended for doctors or medical practitioner only and is not meant
to substitute for the advice provided by a medical professional.
Always consult a physician if you have health concerns. Use and
access of this site is subject to the terms and conditions as set
out in our Privacy Policy and Terms of Use. TAJ GROUP © Copyright
***7 Taj Pharma Group (India),. All rights reserved. PHARMACEUTICAL
B U S I N E S S Note: This site contains medical information that
is intended for doctors or medical practitioner only and is not
meant to substitute for the advice provided by a medical
professional. Always consult a physician if you have health
concerns. Use and access of this site is subject to the terms and
conditions as set out in our Privacy Policy and Terms of Use. ©
Copyright ***7 Taj Pharma Group (India),. All rights
reserved.
| 国: |
India |
| モデル番号: |
-
|
| 离岸价格: |
(交渉可能)
Get Latest Price
|
| ロケーション: |
- |
| 最低注文量の価格: |
- |
| 最低注文量: |
- |
| パッケージの詳細: |
- |
| 納期: |
- |
| 供給能力: |
- |
| 支払いタイプ: |
- |
| 製品グループ : |
- |